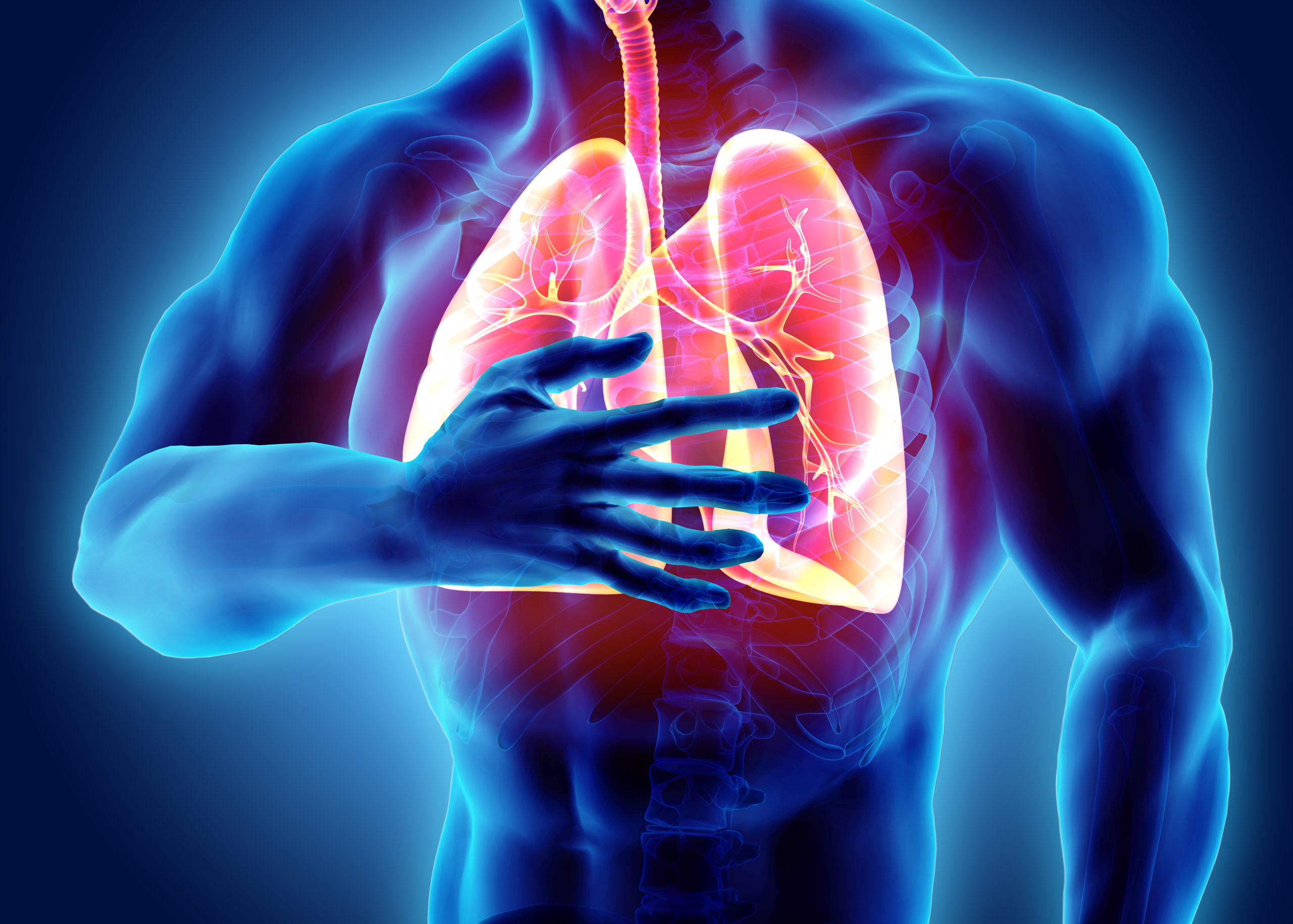
The primary organs of the respiratory system are the lungs. The exchange of carbon dioxide and oxygen in the respiratory system occurs in the alveoli of the lungs. Their job is to take in oxygen and eliminate carbon dioxide as we breathe. Red blood cells receive oxygen from the lungs and transport it to other parts of the body, then the red blood cells collect carbon dioxide and bring it back to the lungs to be exhaled. This exchange of gases is the respiratory system’s way of oxygenating the blood and organ systems. Like other systems, the respiratory system must maintain a balanced pH. Healthy lungs have a pH similar to that of the blood (7.38 – 7.42).
The respiratory system manipulates the flow of carbon dioxide (CO2) in order to help maintain acid-alkali balance. Respiration can activate changes in pH levels within minutes by removing or conserving CO2, directly affecting pH status. When excess acidity is present, bicarbonates will buffer the fluids to try to eliminate the excess CO2. On the other hand, if pH is too alkaline, ventilation is inhibited, and the respiratory rate decreases to try to retain slipping CO2 levels.
Richardson, a researcher from the Journal of Clinical Investigation, studied the effects of blood pH changes and CO2 tension (pCO2 – amount of carbon dioxide gas dissolved in the blood) on healthy subjects. Compared to the control group, it was found that pH levels were significantly altered in groups following acid and base infusions. Acid infusions were accompanied by a significant increase in arterial PCo2 and CO2, whereas alkalizing infusions were associated with reductions in CO2 content.
With that being said, carbon dioxide is imperative for oxygen utilization, but the right balance is required. When there is adequate oxygen-carbon dioxide exchange, the blood will maintain a balanced pH. Oxygen is only promptly released from red blood cells (which are needed to supply oxygen to the tissues), in the presence of sufficient levels of carbon dioxide. If carbon dioxide is lost too quickly, like with mouth breathing or anxiety instead of nose breathing, negative pressure builds in the lungs, and oxygen absorption and utilization is limited. Too much carbon dioxide, like with asthma, can lead to respiratory acidosis. Too little, like with hyperventilation, can lead to respiratory alkalosis. Labored or shallow breathing, which is common in obese people, can result in hypoventilation, meaning too little oxygen taken in and too much carbon dioxide remaining in the blood. This can sometimes cause voluntary hyperventilation to compensate for lack of oxygen, ironically resulting in respiratory alkalosis, a clinical pH imbalance.
When the tissues, organs, and fluids are in a constant state of acidity, which is far more common than not, red blood cells clump together and cannot transport oxygen fluently. This causes cells to suffocate, preventing normal aerobic cellular energy, due to lack of oxygen and improper elimination of carbon dioxide. As acidity increases, mostly from poor diet and lifestyle choices, inflammation and excessive mucus will form. Excessive mucus can compromise the bronchi, or passage ways in the lungs. When this pattern continues, pathogenic material can build up, leaving you much more vulnerable to contracting viruses, infections, repetitive colds, bronchitis, pneumonia, sinusitis, allergies, asthma, etc. A pH balancing anti-inflammatory diet and lifestyle can allow the body to go into a state of homeostasis, working to clear excess mucous, heal the lung tissue, and reverse these adverse conditions As you can see, the respiratory system plays a significant role in maintaining pH balance!
Richardson, D. W., Wasserman, A. J., & Patterson Jr, J. L. (1961). General and regional circulatory responses to change in blood pH and carbon dioxide tension. Journal of Clinical Investigation, 40(1), 31.
